https://doi.org/10.1351/goldbook.O04367
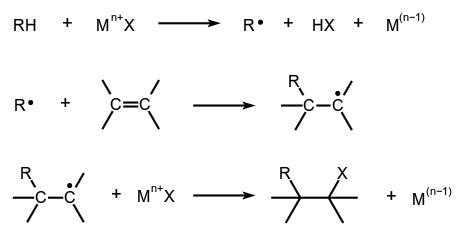
The insertion of a metal complex into a covalent bond involving formally an overall two-electron loss on one metal or a one-electron loss on each of two metals, i.e.