https://doi.org/10.1351/goldbook.H02867
Monocyclic planar (or almost planar) systems of trigonally (or sometimes digonally) hybridized atoms that contain (4n + 2) π-electrons (where n is a non-negative integer) will exhibit aromatic character. The rule is generally limited to n = 0–5. This rule is derived from the Hückel MO calculation on planar monocyclic conjugated hydrocarbons (CH)m where m is an integer equal to or greater than 3 according to which (4n + 2) π-electrons are contained in a closed-shell system. Examples of systems that obey the Hückel rule include: 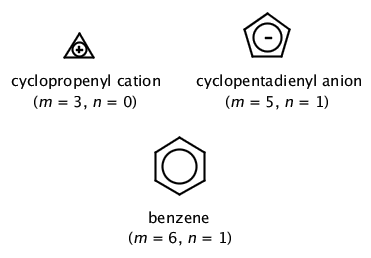 Systems containing 4n π-electrons (such as cyclobutadiene and the cyclopentadienyl cation) are 'antiaromatic'.
Systems containing 4n π-electrons (such as cyclobutadiene and the cyclopentadienyl cation) are 'antiaromatic'.

See also: conjugation, Möbius aromaticity