also contains definitions of: heterovalent hyperconjugation, isovalent hyperconjugation, sacrificial hyperconjugation
https://doi.org/10.1351/goldbook.H02924
In the formalism that separates bonds into σ and π types, hyperconjugation is the interaction of σ-bonds (e.g. C–H, C–C, etc.) with a π network. This interaction is customarily illustrated by contributing structures, e.g. for toluene (below), sometimes said to be an example of 'heterovalent' or 'sacrificial hyperconjugation', so named because the contributing structures contains one two-electron bond less than the normal Lewis formula (electron dot or Lewis structure) for toluene: 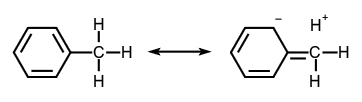 At present, there is no evidence for sacrificial hyperconjugation in neutral hydrocarbons. The concept of hyperconjugation is also applied to carbenium ions and radicals, where the interaction is now between σ-bonds and an unfilled or partially filled π- or p-orbital. A contributing structures illustrating this for the tert-butyl cation is:
At present, there is no evidence for sacrificial hyperconjugation in neutral hydrocarbons. The concept of hyperconjugation is also applied to carbenium ions and radicals, where the interaction is now between σ-bonds and an unfilled or partially filled π- or p-orbital. A contributing structures illustrating this for the tert-butyl cation is:  This latter example is sometimes called an example of 'isovalent hyper-conjugation' (the contributing structures containing the same number of two-electron bonds as the normal Lewis formula). Both structures shown on the right hand side are also examples of 'double bond-no-bond resonance'. The interaction between filled π- or p- orbitals and adjacent antibonding σ* orbitals is referred to as 'negative hyperconjugation', as for example in the fluoroethyl anion:
This latter example is sometimes called an example of 'isovalent hyper-conjugation' (the contributing structures containing the same number of two-electron bonds as the normal Lewis formula). Both structures shown on the right hand side are also examples of 'double bond-no-bond resonance'. The interaction between filled π- or p- orbitals and adjacent antibonding σ* orbitals is referred to as 'negative hyperconjugation', as for example in the fluoroethyl anion: 
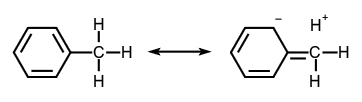


See also: sigma, pi, n-σ*, delocalization