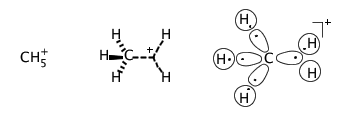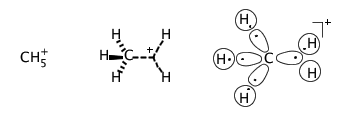https://doi.org/10.1351/goldbook.HT07051
A property of main-group atoms in molecular entities to acquire coordination numbers greater than four (which would comply with the Lewis octet rule). Hypercoordination may be associated with hypervalency, but usually is referred to peculiar atomic centres in the electron-deficient species with multicentre σ-bonding, in which the bonding power of a pair of electrons is spread over more than two atoms. An example of a hypercoordinated atom is the five-coordinate carbon atom in the methanium cation, where three C–H bonds may be regarded as normal two center - two electron bonds and the bonding in the remaining CH2 fragment is governed by the three-centre, two-electron bond. A particular case of a hypercoordinated atom is the hydrogen atom included into a hydrogen bond.